These information suggest that the effect of chemerin for FLS mobility is just not affected by inducing the production of CCL2. Within the RA joints, the pannus tissue migrates and invades the cartilage and bone, which contribute to damaging these structures. FLSs would be the predomi nant cell sort in pannus tissue, specially at the pannus cartilage junction. FLSs retrieved from synovial tis sues directly result in cartilage degradation when cocul tured with macrophages in vitro, suggesting that FLS migration and invasion play a central function in pannus tissue connected cartilage degradation in RA. Moreover, our benefits show that chemerin enhances MMP 3 pro duction from RA FLSs, which can be a proteolytic enzyme with cartilage degradation properties. Collectively, our outcomes suggest that chemerin plays a vital part in cartilage destruction by way of FLS activation.
The present outcomes show that chemerin enhances the activation of ERK1 2, p38MAPK and Akt, but not of JNK1 2 and NF B, in FLSs. Furthermore, pretreatment having a specific inhibitor article source of MEK, p38MAPK, and PI3K suppressed chemerin induced IL six production, and p38 MAPK and PI3 kinase inhibitor reduced RA FLS cell motility. These final results suggest the involvement of each the MAPK and PI3K Akt pathways in chemerin induced IL 6 produc tion by RA FLSs. The p38MAPK and PI3K Akt path methods are also involved in cell motility induced by chemerin. Chemerin activated macrophage adhesion to fibronectin by activation of p38MAPK and PI3K Akt signaling pathway. These outcomes suggest that che merin activates macrophages too as FLSs in RA synovium.
Conclusions Our results recognize the critical function of chemerin in the activation of FLSs in RA synovium, suggesting that chemerin and ChemR23 interaction may possibly play a function in the pathogenesis of RA. Introduction selleckchem MG-132 Osteoarthritis would be the most typical worldwide articular disease and affects a large quantity of adults. It outcomes from articular cartilage failure induced by the interactions of genetic, metabolic, biochemical, and bio mechanical components with the secondary components of inflammation. The processes underlying OA involve interactive degradation and repair systems in cartilage, bone, plus the synovium. It is also now believed that syno vial inflammation and the production of proinflammatory or destructive mediators from the OA synovium are critical for the progression of OA.
Synovial tissues from individuals with early signs of OA show infiltrations of macrophages that exhibit an activated phenotype and produce proinflammatory cytokines, primarily 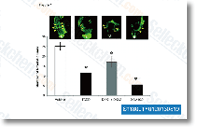 interleukin 1b and tumor necrosis element a. Macrophage derived IL 1b and TNF a are expected for the release of matrix metalloproteinases from the synovium that should eventually degrade cartilage tissues. In addition, it has been observed that macrophages mediate osteophyte formation and fibrosis in the early stages of experimentally induced OA.
interleukin 1b and tumor necrosis element a. Macrophage derived IL 1b and TNF a are expected for the release of matrix metalloproteinases from the synovium that should eventually degrade cartilage tissues. In addition, it has been observed that macrophages mediate osteophyte formation and fibrosis in the early stages of experimentally induced OA.
Blogroll
-
Recent Posts
- Multi-chemical examination joined with chemometrics for you to define PDO along with PGI Italian oatmeal.
- Novel combination peptide-mediated siRNA delivery using self-assembled nanocomplex.
- Early on along with Mid-Term Implications in the COVID-19 Crisis about the Physical, Behavioral as well as Psychological Health associated with The medical staff: The actual CoPE-HCP Study Method.
- Long-term Impulsive Pneumoperitoneum along with Pneumatosis Cystoides Intestinalis of the Minor and major Bowel.
- Nebulized Micafungin Strategy for Scopulariopsis/Microascus Tracheobronchitis throughout Bronchi Hair treatment People.
Archives
- April 2024
- March 2024
- February 2024
- January 2024
- December 2023
- November 2023
- October 2023
- September 2023
- August 2023
- July 2023
- June 2023
- May 2023
- April 2023
- March 2023
- February 2023
- January 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- March 2022
- February 2022
- January 2022
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- October 2020
- September 2020
- August 2020
- July 2020
- June 2020
- May 2020
- April 2020
- March 2020
- February 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- August 2019
- July 2019
- June 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- October 2016
- September 2016
- August 2016
- July 2016
- June 2016
- May 2016
- April 2016
- March 2016
- February 2016
- January 2016
- December 2015
- November 2015
- October 2015
- September 2015
- June 2015
- May 2015
- April 2015
- March 2015
- February 2015
- January 2015
- December 2014
- November 2014
- October 2014
- September 2014
- August 2014
- July 2014
- June 2014
- May 2014
- April 2014
- March 2014
- February 2014
- January 2014
- December 2013
- November 2013
- October 2013
- September 2013
- August 2013
- July 2013
- June 2013
- May 2013
- April 2013
- March 2013
- February 2013
- January 2013
- December 2012
- November 2012
- October 2012
- September 2012
- August 2012
- July 2012
- June 2012
- May 2012
- April 2012
- March 2012
- February 2012
- January 2012
Categories
Tags
Anti-CD4 Anti-CD4 Antibody anti-CD4 monoclonal antibody Anti-CD44 Anti-CD44 Antibody Anti-PTEN Anti-PTEN Antibody BMS512148 CD4 Antibody CD44 Antibody CHIR-258 CT99021 custom peptide price cytoplasmic DCC-2036 DNA-PK Ecdysone Entinostat Enzastaurin Enzastaurin DCC-2036 GABA receptor GDC-0449 GSK1363089 Hyaluronan ITMN-191 kinase inhibitor library for screening LY-411575 LY294002 MEK Inhibitors mouse mTOR Inhibitors Natural products oligopeptide synthesis organelles PARP Inhibitors Peptide products Pfizer proteins PTEN Antibody small molecule library solid phase Peptide synthesis Sunitinib Sutent ZM-447439 {PaclitaxelMeta
